Global Preeclampsia Market to Grow from USD 1.36 Billion in 2025 to USD 3.18 Billion by 2035, Recording 8.9% CAGR
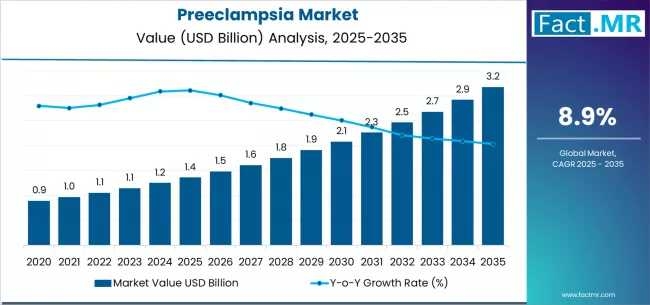
The global preeclampsia market is entering a decade of rapid diagnostic evolution, projected to rise from USD 1.36 billion in 2025 to USD 3.18 billion by 2035. According to a definitive market outlook by Fact.MR, the sector is advancing at a robust 8.9% CAGR, driven by a fundamental shift toward early-first-trimester screening and the widespread clinical adoption of biomarker-based blood assays.
As maternal healthcare systems worldwide prioritize the reduction of pregnancy-related morbidity, preeclampsia testing has transitioned from reactive symptom management to proactive risk stratification. The market is currently dominated by Blood Tests, which hold a 59.0% share, underpinned by the clinical gold standard: the sFlt-1/PlGF ratio assay.
Get Access Report Sample : https://www.factmr.com/connectus/sample?flag=S&rep_id=11601
Quick Stats: Preeclampsia Market at a Glance
- Market Value (2025E): USD 1.36 Billion
- Projected Value (2035F): USD 3.18 Billion
- Forecast CAGR:9% (2025–2035)
- Leading Test Type: Blood Tests (59.0% share)
- Primary End-User: Diagnostic Centers (35.0% share)
- Growth Hotspots: India (10.2% CAGR) and China (9.8% CAGR)
A Decade of Growth: Two Distinct Phases
The market’s expansion is characterized by a structural shift in how clinicians identify at-risk pregnancies:
Phase 1: 2025–2030 (The Awareness Wave)
The market is expected to reach USD 2.13 billion by 2030. This period is defined by the integration of placental growth factor (PlGF) and uterine artery Doppler analysis into routine prenatal care. Government-funded programs in middle-income nations are currently expanding screening capacities to include these biochemical parameters.
Phase 2: 2030–2035 (The Innovation Wave)
Growth will accelerate to USD 3.18 billion, fueled by the introduction of multiplex assay systems and point-of-care (POC) microfluidic devices. This era will see the integration of Artificial Intelligence (AI) and longitudinal proteomics, allowing for personalized risk prediction well before clinical symptoms manifest.
Segmental Spotlight: Blood Tests and Universal Screening
Blood Tests remain the dominant methodology, with the sFlt-1/PlGF ratio alone representing 31.0% of the segment. Recent FDA clearances—such as Roche’s Elecsys sFlt-1/PlGF ratio and the commercial launch of Trinity Biotech’s PreClara test—have provided the clinical confidence and reimbursement pathways necessary for mass-market adoption.
Universal Screening applications currently account for 40.0% of demand. Leading obstetricians are increasingly advocating for first-trimester screening protocols (representing 22.0% of this segment) to enable early intervention strategies, such as low-dose aspirin regimens, which can significantly alter the disease trajectory.
Regional Performance: Asia-Pacific Leading Market Velocity
While the United States (8.9% CAGR) maintains clinical leadership through sophisticated diagnostic deployment in urban medical centers, the Asia-Pacific region is the world's fastest-growing corridor.
|
Country |
Projected CAGR |
Primary Growth Catalyst |
|
India |
10.2% |
Government-led maternal screening rollouts and low-cost biomarker adoption. |
|
China |
9.8% |
Hospital digitization and regulatory approval of rapid urine/blood tests. |
|
USA |
8.9% |
High awareness and FDA-cleared biomarker risk stratification systems. |
|
Germany |
8.4% |
Universal screening integration under stringent EU clinical guidelines. |
|
UK |
8.1% |
NICE-endorsed PlGF test adoption across NHS frameworks. |
Strategic Drivers & Industry Challenges
Market Drivers:
- Clinical Modernization: Growing reliance on biomarkers over traditional blood pressure monitoring for accurate diagnosis.
- Regulatory Support: NICE (UK) and FDA (USA) endorsements are standardizing screening guidelines globally.
- Digital Integration: The use of Laboratory Information Systems (LIS) and AI to track longitudinal biomarker changes throughout pregnancy.
Industry Restraints:
- Implementation Complexity: Variations in testing accuracy across different laboratory settings can hinder universal validation.
- Supply Chain Volatility: The consistent availability of specialized biomarker reagents remains a concern for large-scale screening programs.
Competitive Landscape
The preeclampsia market is a high-tech arena featuring global giants such as Roche, Thermo Fisher Scientific, Siemens Healthineers, and Siemens Healthineers, alongside specialized innovators like Sera Prognostics and Trinity Biotech. Competitive differentiation is now being driven by the development of point-of-care platforms and multiplex assays that can deliver results in minutes rather than hours.
Analyst Opinion:
"We are witnessing the transition of preeclampsia from a 'silent killer' to a manageable condition. The convergence of microfluidic POC devices and AI-driven predictive analytics is allowing clinicians to identify at-risk patients in the first trimester. Companies that can bridge the gap between high-complexity lab results and rapid bedside testing will define the next decade of maternal health." — Principal Consultant, Fact.MR
Interactive Next Step
Would you like me to prepare a technical comparison between the sFlt-1/PlGF ratio assay and newer proteomic biomarker panels to help you evaluate the next generation of diagnostic investments?
To View Related Report:
ETO Manufacturing Market https://www.factmr.com/report/eto-manufacturing-market
Sanitizer Test Strips Market https://www.factmr.com/report/sanitizer-test-strips-market
Digital Transformation in Healthcare Market https://www.factmr.com/report/digital-transformation-in-healthcare-market
Anesthesia Machine Market https://www.factmr.com/report/96/anesthesia-machines-market
- Contact Us -
11140 Rockville Pike, Suite 400, Rockville,
MD 20852, United States
Tel: +1 (628) 251-1583 | sales@factmr.com
About Fact.MR
Fact.MR is a global market research and consulting firm, trusted by Fortune 500 companies and emerging businesses for reliable insights and strategic intelligence. With a presence across the U.S., UK, India, and Dubai, we deliver data-driven research and tailored consulting solutions across 30+ industries and 1,000+ markets. Backed by deep expertise and advanced analytics, Fact.MR helps organizations uncover opportunities, reduce risks, and make informed decisions for sustainable growth.
- Sports
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Jeux
- Gardening
- Health
- Domicile
- Literature
- Music
- Networking
- Autre
- Party
- Shopping
- Theater
- Wellness


